And yet, the mouse still sits at the center of the drug development pipeline. For nearly a century, mice (and to a lesser extent, dogs, pigs, and nonhuman primates) have served as the go-to models for predicting how new treatments will work in humans. It’s a practice rooted in convenience, regulation, and a lack of better options. But it may also be holding medicine back.
It’s a system that’s expensive, inefficient, and deeply reliant on an approach that doesn’t always translate. It often takes more than $2.5 billion and over a decade to bring a single drug to market, and most still don’t make it. Mouse models are convenient, but they’re also limited. Many therapies that work in rodents go on to disappoint in humans. And perhaps more troubling is the possibility that some experimental compounds that failed in mice might work in humans if only we had a better way to test them.
Now, a UC Irvine startup is offering an alternative.
Aracari Biosciences has developed a new kind of drug testing platform—an organ-on-a-chip that mimics the structure and function of living human tissue. It was co-founded by Christopher Hughes (UC Irvine professor of molecular biology and biochemistry), Steven George (formerly at UC Irvine and currently a professor of biomedical engineering at UC Davis), and Abraham Lee (Chancellor’s Professor of biomedical engineering and mechanical and aerospace engineering at UC Irvine). The company is pursuing two key ambitions: to improve how new medicines are developed and to test treatments on a patient’s own cells or tissues before a prescription is written.
“There was a huge gulf that needed to be filled,” says Hughes. “That’s where the microphysiological systems, or the organ-on-a-chip, come in. They fill in that middle ground between simple cell cultures and animal models.”

Much of Aracari’s early momentum came from the broader UC Irvine innovation ecosystem. Hughes received a Proof of Product (PoP) grant from UC Irvine Beall Applied Innovation, which helped fund initial steps toward commercialization. The company was also one of the early tenants of University Lab Partners (ULP), a UC Irvine-affiliated wet lab incubator that provided access to lab space, infrastructure and a collaborative startup environment.
“We were one of the first companies in the ULP space. It was a great setup. Everything we needed to get moving was right there,” says Hughes.
That combination of translational funding and physical resources allowed the founders to accelerate development and refine their technology before moving into independent space.
A Model More Like Us
Nearly every cell in the body sits within a hair’s width of a blood vessel. This is how oxygen, nutrients, and medications reach their targets and how waste gets carried away. Without the vascular system, a model offers only a partial picture of how treatment actually works.
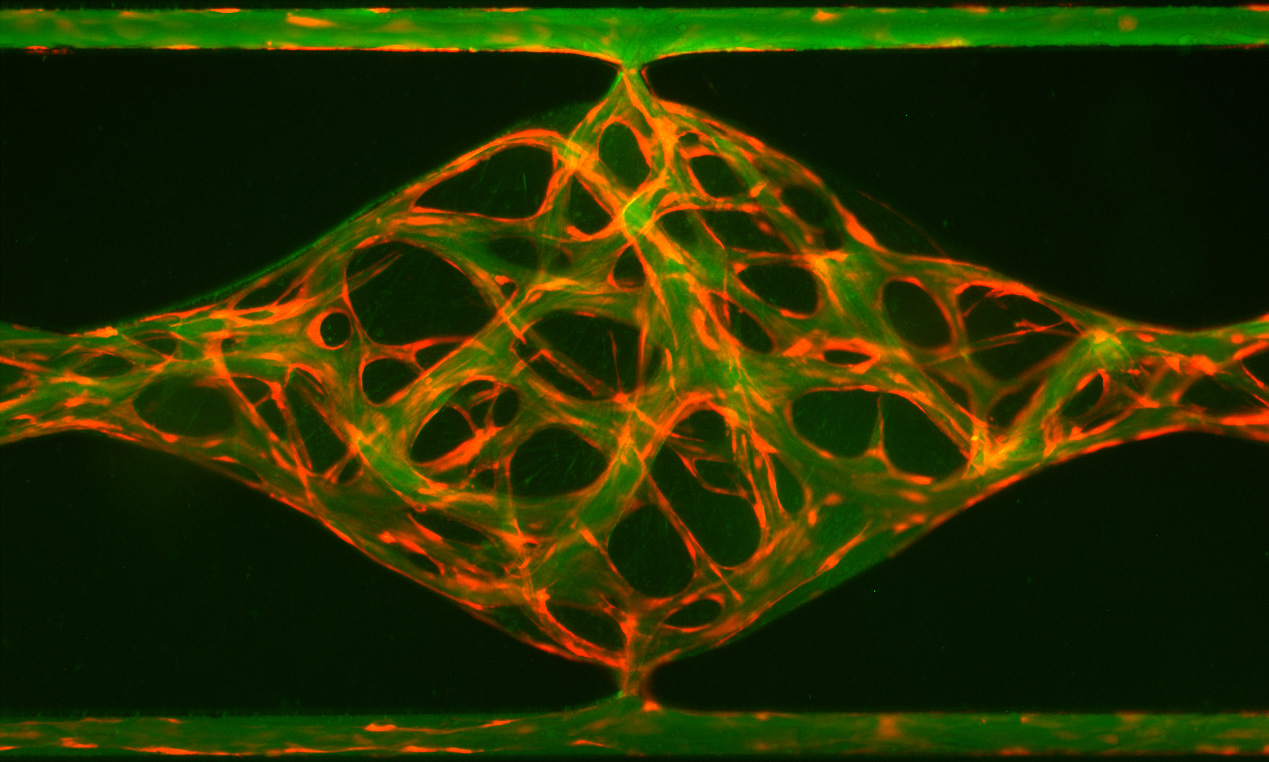
Aracari’s microfluidic system uses real human cells to recreate tiny, living 3D tissues, complete with a functioning vascular system. It’s like a miniaturized organ in a small dish, with channels for blood flow and just enough complexity to capture how cells interact with drugs under realistic conditions.
“Because the platforms are made with human cells, the results are far more reflective of actual patients,” says Hughes. “You don’t have to wonder if it’ll behave the way it does in a mouse.”
Unlike traditional 2D cell cultures or animal models, Aracari’s chips offer a close approximation of human physiology. The result is a tool that makes drug development less of a guessing game.
“Because the platforms are made with human cells, the results are far more reflective of actual patients”
– Christopher Hughes, Founder, Aracari Biosciences and UC Irvine Professor of Molecular Biology and Biochemistry
Initially, Aracari’s team ran tests in-house on behalf of its clients. Today, while that work continues, the company is also expanding access by shipping its organ-on-a-chip platform directly to customers for them to run their own tests. Some use it to decide which therapies are worth pursuing. Others turn to the system when unexpected toxicity arises in animal studies or early trials.
Letting clients run their own tests brought the platform’s value into sharper focus. When Aracari began beta-testing shipped platforms, client enthusiasm confirmed they were onto something.
“Their results were so good they wanted to start paying us right away,” Hughes says. “They told us, ‘We don’t want this to be a free test anymore. We want to make sure we can keep working with you.’ That’s when we realized this had real commercial potential.”
As Aracari expands its reach, Lee believes the implications go beyond just improving models. They could reshape how drugs are discovered.
“I believe this system has the potential to be a mainstay in screening drug compounds and discovering drugs that are safe and potent,” says Lee, whose expertise in microfluidics helped give Aracari’s platform its unique structure and control.
Today, Aracari counts seven of the world’s top ten pharmaceutical companies among its clients, a sign of how eager the industry is for more predictive, human-relevant models.
Hughes estimates that roughly 75% of Aracari’s clients are focused on oncology—and for good reason. Cancer drug development remains one of the riskiest bets in the pharmaceutical industry. Despite billions invested and promising results in animal models, nearly 90% of cancer drugs that enter clinical trials ultimately fail to reach the market. For every ten candidates that enter the pipeline, only one is likely to succeed.
“It’s pretty easy to cure cancer in mice, but very few of those actually translate to the clinic,” Hughes says.
About 20% of Aracari’s other clients are investigating toxicity, while the remaining span a wide range of fields, from lung fibrosis to oral drug absorption. In short, Aracari’s platform isn’t limited to one disease area or function. It’s engineered for a wide range of applications.
“We are disease agnostic,” Hughes says. “You can use our platform for anything. That’s the beauty of it. It’s very adaptable.”
Speculation Becomes Science
If the first phase of Aracari was proving its platform worked, the next phase is about scaling its impact. The company is now focused on expanding its customer base and bringing new tissue models to market.
One model in development is a platform of the blood-brain barrier, the tight network of vessels that protects the brain but also blocks many therapies from getting through.
“Everybody’s chasing a good model of the blood-brain barrier,” says Hughes. “It’s one of the biggest challenges in drug delivery.
Meanwhile, Aracari’s long-term ambition is to transform how we approach precision-based care. Their idea is to take a sample of a patient’s cells, grow it on a chip, and test multiple drugs to find the one that works. The idea is to prescribe medicine not based on what’s likely to work for someone statistically similar, but on what demonstrably works for you.
It’s a concept Hughes calls “two-way personalized medicine.” “Personalized medicine” often means tailoring treatments based on a person’s genetic makeup or how others with similar genes have responded. But that approach still leans heavily on inference. It functions more like a one-way mirror.
Aracari’s vision turns that one-way mirror into more of a two-way window, one that reveals, for example, how a patient’s tumor responds to treatment. Instead of predicting a response, it generates one. Physicians could move from educated guesses to observed results.
That shift has profound implications. For patients, it promises less trial and error, and more clarity in moments when the stakes are high and guidance is needed most. For clinicians, it introduces a new kind of diagnostic that doesn’t just say what kind of tumor it is, but how it’ll likely respond. And for drug developers, it could offer a powerful feedback loop, or a way to see, early and concretely, which compounds work for which patients.
Unfortunately, that future isn’t here quite yet. Clinical adoption will take time, and current regulatory frameworks weren’t designed with tumor-in-a-chip prescriptions in mind. But the foundation is in place. The organ-on-a-chip isn’t just a model of biology—it’s a model of what individualized treatment could become.
What if we didn’t need to rely on mice at all? What if we could test drugs on real human tissue before the first clinical trial ever begins? What if, instead of trial-and-error, we knew?
These questions used to live in the realm of speculation. Now, Aracari is working to turn those what-ifs into real-world answers.