The result of more than 15 years of research focused on unmet clinical needs across valvular heart disease and heart failure comes to fruition in the UCI Startup, ValVention, a company devoted to mitigating structural heart disease through the development of next-generation transcatheter heart valves and minimally invasive cardiac support technology.
Founded by UCI Professor, Arash Kheradvar, M.D., Ph.D., FAHA, ValVention has made leaps toward bringing three different products to the marketplace including; FoldaValve™, HelixCardia™, and A-Valve™.
- FoldaValve™ is a next generation Transcatheter Aortic Valve Replacement (TAVR) system that is capable of fully repositioning of the valve during implantation procedure and provide retrieval option, as needed. At its 14-Fr delivery size, FoldaValve™ uniquely protects the leaflets from being stent-crimped during delivery, and therefore, minimizes the leaflet damage. FoldaValve™ delivery system, Ingenuity™, is distinctively equipped with an intravascular ultrasound (IVUS) system that guides the delivery and implantation of the valve.
- HelixCardia™ is a percutaneously implantable cardiac support device inspired by the concept of helically arranged myocardial fibers. It has no direct contact with circulating blood, so it requires no blood-thinning medications, which eliminates risks of stroke and bleeding. HelixCardia™ is internally powered, transcutaneously rechargeable and is aimed to help patients with advanced heart failure that may never receive a heart transplant and prefer not to receive a ventricular assist device due to its lifestyle limitations.
- A-Valve™ is the only modular transcatheter mitral valve system. It is made to accommodate different patients’ anatomies and diseases by offering different sizes of atrial stent and valve coupled to each other in the Cath lab based on the physician’s decision. Currently there is no FDA-approved transcatheter mitral valve. This is because of the variation among patients which makes it challenging for a device to work for all patients. A-Valve uniquely provides flexibility in different valve components to be assembled in the Cathlab by the operating physician to match different patients’ anatomies and diseases.
“Unmet needs represent an innovator’s best opportunity to impact outcomes; it’s an opportunity to improve an existing product or process… we might see an entirely new product or process developed, or when we start putting different disciplines together solutions never thought possible start to surface,” says UCI’s Chief Innovation Officer, Errol Arkilic.
Dr. Kheradvar is a big believer of collaboration and research within the University setting. He emphasizes that: “As a university researcher you have access to many resources that would otherwise be unavailable or costly to you. An academic institution allows researchers to get involved in different types of research and acts as an umbrella to support them. Us as the professors are very fortunate to have the opportunity to work with many brilliant students, postdocs, and researchers. Our lab progress is the result of working and collaborating with a diverse group of experts who view the problems and solutions from various angles.”
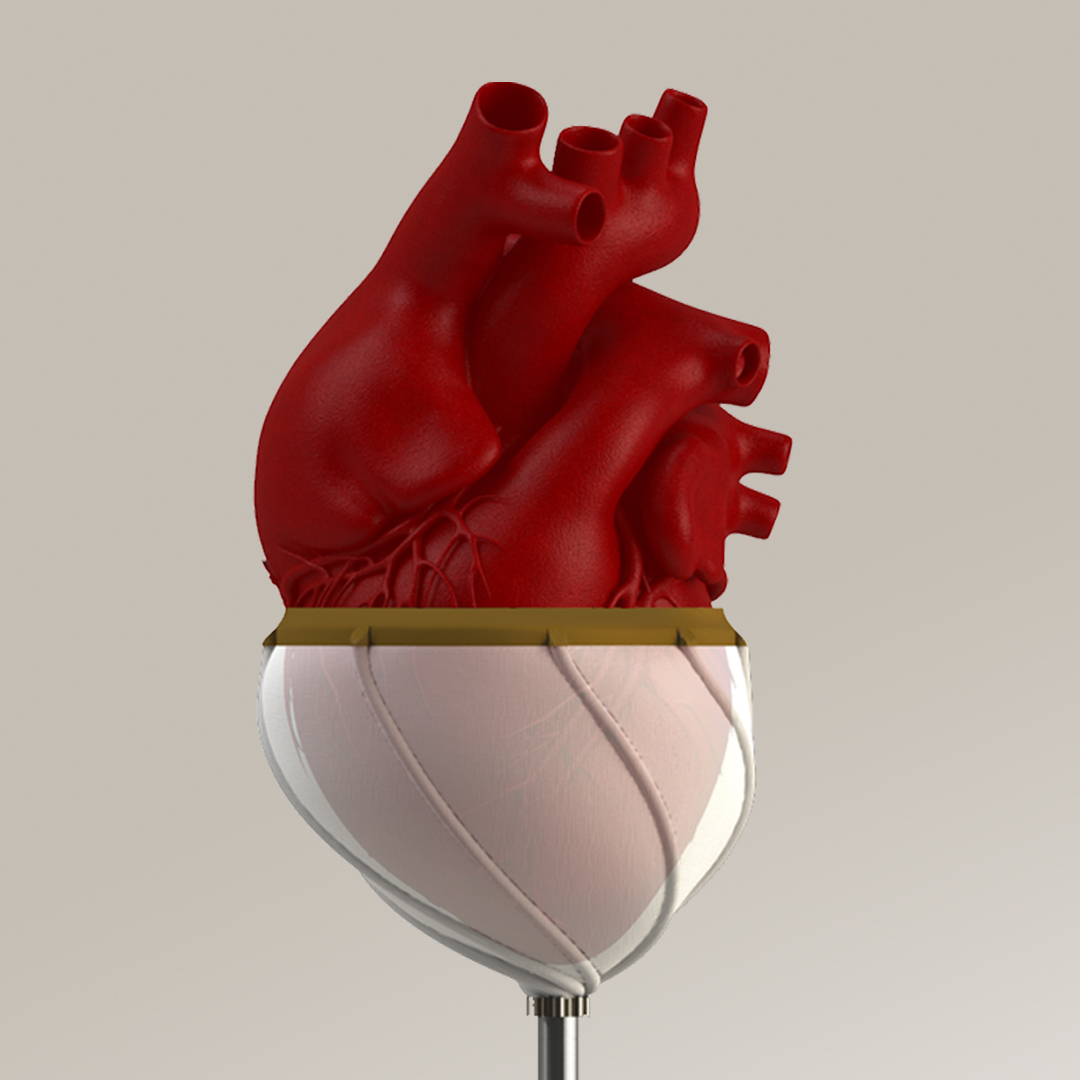
Two of the three technologies that ValVention is licensing from UCI are the recipient of Proof of Product (PoP) funds awarded by the UCI Beall Applied Innovation to fill the commercialization gap. The PoP grants have been highly competitive to get given that their applications are reviewed by a multidisciplinary and interdisciplinary group of experts. PoP grants provide UCI researchers with the means to perform exclusive research needed to move forward a technology towards commercialization, and help discoveries closer to the marketplace. It does so by helping “de-risk” the technology and more clearly identifying its potential trajectory, which makes the technology and team behind it more attractive for investment.
For Applied Innovation, PoP serves as an indicator for identifying UCI researchers who seek to make a greater impact to the society by accelerating the advancement of their knowledge into a technology that can be commercialized.
“Early identification of research with commercialization potential can make a world of difference to its ultimate impact. When you bring varied viewpoints and voices to an emerging idea, the paths it can take are broad. As that idea becomes more and more specific, pathways start to fall off and the relevance of its application in other areas start to go with them,” says Arkilic.
“We don’t seek to tell our researchers how to research, or what to research, we seek to amplify the potential of their research,” Arkilic adds.
Dr. Kheradvar was an early adopter of PoP (now in its 8th cycle), receiving funds from both round 3 and round 6 of the donor-backed program. This early adoption helped advance ValVention’s A-Valve™ and HelixCardia™ products towards commercialization. In the case of HelixCardia™, the PoP program provided valuable feedback to help influence trajectory before ultimately funding the project. In both cases, funding was used for feasibility animal studies that have significantly moved the technologies forward.
In addition to PoP funding, ValVention received an SBIR grant from NHLBI, as well as an NIH grant to advance its innovative FoldaValve™ Technology.
By creating a startup where the knowledge developed in his lab is being commercialized, Dr. Kheradvar aims to positively impact the life of millions of patients suffering from heart failure and heart valve disease.
UCI Researchers seeking proof of product funding and support should visit innovation.uci.edu/pop-grants to learn more.
